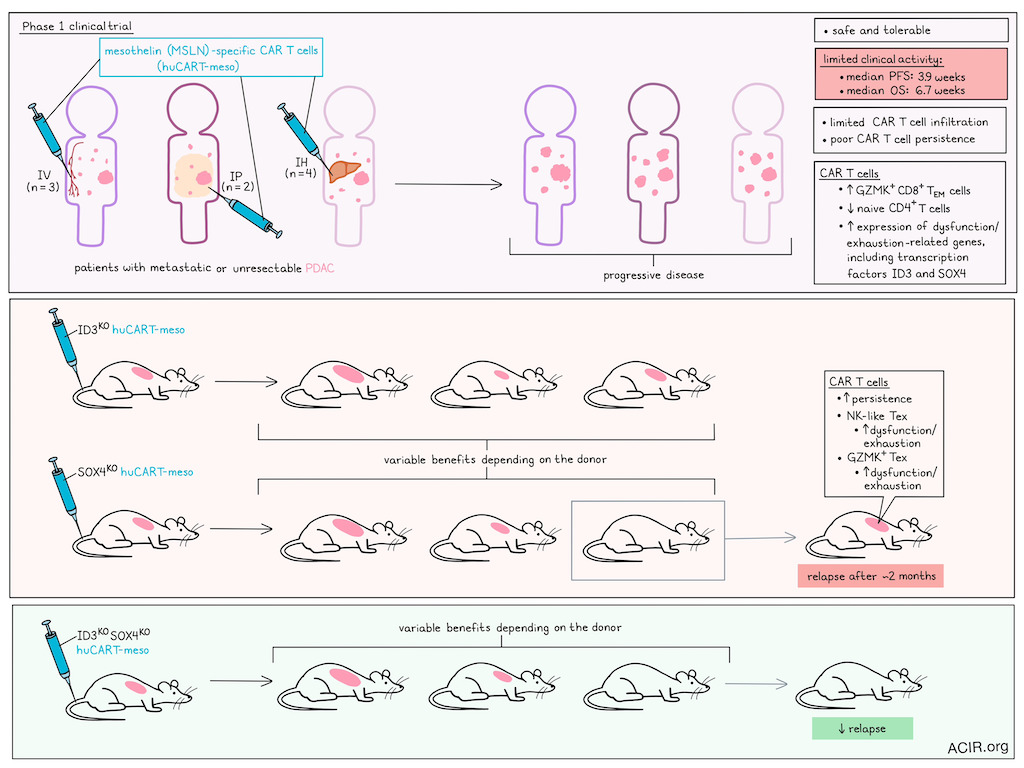
Characterized by very poor outcomes and an immune-cold tumor phenotype, pancreatic ductal adenocarcinoma (PDAC) poses a major challenge for immunotherapy treatment. Aznar and Good et al. performed a phase I study assessing the safety and feasibility of a mesothelin (MSLN)-targeting CAR T cell product (huCART-meso) and performed subsequent analyses of the infused cells, as well as correlative mouse studies to determine treatment resistance mechanisms. Their results were recently published in Cell Reports Medicine.
The study included patients with unresectable or metastatic PDAC, who had received at least one prior line of systemic treatment. CAR-T were provided either via intravenous (IV; n=3), intraperitoneal (IP; n=2), or intrahepatic (IH; n=4) delivery (patients with IP and IH delivery were eligible for follow-up IV delivery). One patient in the IV cohort had pulmonary toxicity, potentially due to first-pass effect, which prompted switching of delivery to IP and IH to mitigate this complication. Treatment was tolerated and generally safe, with no related adverse effects above grade 3.
The clinical activity of huCART-meso was limited, and all patients died before long-term follow-up was reached. On day 28, one patient had stable disease as the best response, but progression was noted at month 3, and all other patients had progressive disease. The median overall survival from infusion date was 6.7 weeks, and the median progression-free survival was 3.9 weeks.
Peak levels of huCART-meso cells in circulation were similar between administration routes, but cells persisted longer in the blood after IV administration. HuCART-meso cells were detected in biopsy and peritoneal fluid between days 13 and 26 in patients from all 3 cohorts. Evaluable tumor biopsies had limited CD3+ T cell infiltration at baseline and at day 14 post-infusion, confirming an immune-cold environment. In tissues and blood, CAR downregulation was observed, while MSLN protein levels on EPCAM+ viable tumor cells decreased in post-infusion samples from 2 of 3 evaluated patients. Despite the local delivery into the peritoneal fluid, either low levels or no CAR-expressing T cells were detected in ascites fluid of IP-treated patients at day 7, suggestive of low persistence of the CAR T cells.
ScRNAseq of CD3+ T cells obtained from peritoneal biopsies on days 7 and 26 post-CAR-T infusion included Treg, naive, and effector memory (Tem) CD4+ T cell populations, along with memory, Tem, GZMK+ Tem, terminally-differentiated Tem, and NK-like CD8+ T cell populations. In two patients, 380 and 157 CAR+ cells were detected in the peritoneal fluid. Comparing the CAR+ to the CAR- T cells showed enrichment of the GZMK+ CD8+ Tem cluster and depletion of the naive CD4+ cluster among CAR+ cells.
Previously, the researchers identified a 30-gene dysfunction signature in huCART-meso cells chronically exposed to tumor antigen in vitro. Among CAR+ cells, 19 of the 30 genes were highly expressed in the patient samples, including transcription factors SOX4 and ID3, as well as other dysfunction genes (CTLA4, TIGIT, PDCD1).
Aznar and Good et al. then moved to murine models to evaluate whether ID3KO or SOX4KO might improve the cytotoxicity of huCART-meso cells from healthy donors. The cells were not different from WT cells in terms of viability, population doublings, or cell volume. In a murine xenograft model, suboptimal doses of WT or KO CAR T cells were IV injected, and the KO groups had improved responses for some donors, but there was extensive variation in the observed benefit. The SOX4KO performed better than the ID3KO CAR-T and was further evaluated. The researchers assessed various factors for correlation with in vivo responses (e.g., cell source, total cells, population doublings, surface CAR expression, age, gender) but found no differences. The memory phenotype of the cells was also not associated with in vivo responses.
Some of the mice that initially cleared tumors relapsed at the primary tumor site, starting ~2 months after CAR-T infusion. To determine which factors impact this therapeutic failure, relapsing tumors were analyzed. The KO persisted on the CAR T cells, and TILs were abundantly present, suggesting relapse was not due to lack of persistence. Both the WT and SOX4KO CAR-T-treated mice had reduced levels of surface CAR compared to the infusion product, though the level of CAR+ cells was higher in tumors treated with SOX4KO CAR-T. scRNAseq of CD8+ T cells from the relapsing tumors showed no major differences in cluster distribution. Two main T cell subsets were present – an NK-like T cell exhaustion (NK-like Tex) subset with high levels of NK receptor genes and genes in the 30-gene dysfunction signature, and a GZMK+ T cell exhaustion (GZMK+ Tex) subset, which had high levels of GZMK and EOMES. All clusters highly expressed exhaustion markers and lacked memory markers.
Since both ID3 and SOX4 were upregulated in the exhausted CAR cellsT, the researchers then created double knockouts, aiming to improve antitumor responses. The double-KO CAR-T had similar proliferation, cell volume, and viability during expansion, and there were no differences in the T cell subset distribution. Most of the cells had central or effector memory phenotypes. Treatment with suboptimal doses in AsPC-1 tumor-bearing mice showed similar antitumor efficacy as the single-KO CAR-T.
To determine whether the double-KO CAR-T could prevent relapse after initial response, mice were treated with optimal doses and monitored for relapses. While mice treated with WT or single-KO CAR-T relapsed at early time points, starting after 2 months, the double-KO-treated mice had fewer, delayed, or no relapses for the study period, and the mice that did relapse had smaller tumor volumes. Overall, mice treated with double-KO CAR-T had increased relapse-free survival compared to WT CAR-T-treated mice.
Together, this study showed that while treatment with huCART-meso in patients with advanced PDAC was safe, it was not effective. Some reasons for the lack of efficacy were found to be the limited persistence and expansion of CAR T cells in the tumor. Additionally, CAR-T obtained a dysfunctional phenotype, which might be overcome by double KO of the exhaustion-related transcription factors ID3 and SOX4. If this approach is found to be safe for patient treatment, this may aid in improving the efficacy of CAR-T treatment in difficult-to-treat, immune-cold solid tumor types such as PDAC.
Write-up by Maartje Wouters, image by Lauren Hitchings




