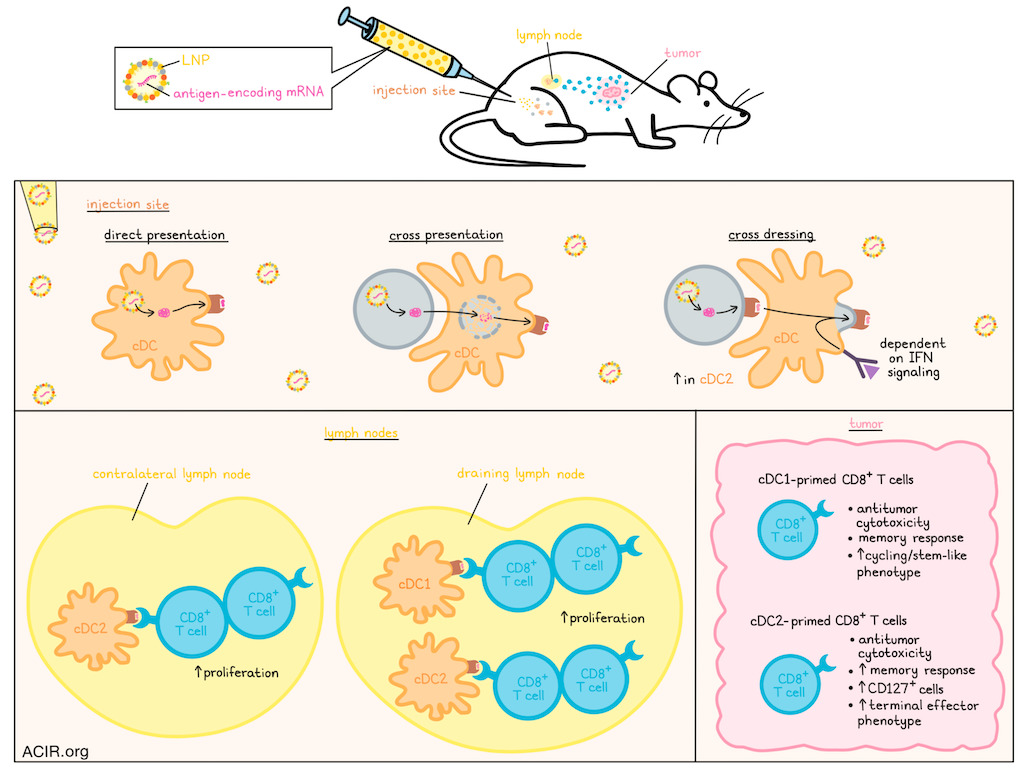
cDC1s play essential roles in CD8+ T cell priming in response to protein- and DNA-based vaccination. However, whether mRNA vaccines function via a similar mechanism remains unknown. In a recent Nature publication, Jo et al. uncovered the mechanisms of CD8+ T cell priming induced by mRNA-LNP vaccines.
The researchers began by assessing OT-I T cell proliferation after intramuscular injection of OVA mRNA-LNPs. After two days, OT-I cells proliferated to comparable extents in spleen, blood, and lymph nodes (LNs). Even at a 100-fold reduction in dose, OT-1 cells proliferated at distant sites, suggesting priming in lymphoid tissue. To determine whether stimulation of OT-1 cells in distal tissues (spleen and blood) involved migration of OT-I cells activated at the injection site or draining LN (dLN), mice were treated with FTY720 prior to vaccination to prevent T cell egress. After treatment, OT-I proliferation persisted in the spleen and LNs, suggesting local antigen presentation.
To determine whether muscle cells or macrophages could serve as antigen-presenting cells (APCs) to prime T cells at the injection site, spleens were removed, and CD62L was blocked to prevent naive T cell infiltration into LNs. This treatment reduced OT-I expansion after vaccination, suggesting that OT-I cells must enter lymphoid organs for priming.
The researchers examined which APCs were responsible for T cell priming by collecting cDC1s, cDC2s, and B cells from dLNs and contralateral LNs (cLNs) on day 2 after vaccination. They tested these for their ability to induce OT-I proliferation. cDC1s and cDC2s from dLNs and cDC2s from cLNs induced proliferation, but B cells did not.
Next, CD8+ T cell responses were assessed in mice lacking cDC1s (Δ32 mice) or cDC2s (Δ1+2+3 mice). When mice were vaccinated with an OVA cDNA or OVA protein vaccine, only the WT and cDC2-deficient mice elicited T cell responses, confirming the necessity of cDC1. The OVA mRNA-LNP vaccine, in contrast, also induced T cell responses in cDC1-deficient mice. Similar results were found with a tumor neoantigen mRNA. Therefore, mRNA vaccines induce CD8+ T cell responses via cDC1s and cDC2s as APCs.
To validate the cDC requirement for T cell priming, CD11c-DTR mice were treated with diphtheria toxin to deplete cDCs, and were vaccinated with OVA mRNA-LNP, which nearly abrogated T cell responses. Similar effects were observed in a CD11c-DTR-to-SJL bone marrow (BM) chimera mouse model, in which cDCs, but not macrophages or monocytes, are depleted. Further, Δ32 × Δ1+2+3 mice lacking cDC1 and monocytes showed normal specific T cell expansion in response to the vaccine, confirming monocytes were not required for cDC1-independent T cell priming.
Jo et al. previously found that vaccine-derived peptides could be loaded onto MHC-I molecules via direct transfection of APCs or cross-presentation. WDFY4 is essential for cross-presenting cell-associated and immune-complex antigens. However, CD8+ T cell responses induced by mRNA-LNP were similar between WT and Wdfy4-/- mice, indicating cross-presentation is not a primary mechanism.
This led the researchers to cross-dressing, an antigen-presentation pathway in which peptide-MHC-I complexes are transferred from donor cells to cDCs before T cell activation. Since this mechanism bypasses the requirement for MHC-I expression by cDCs, mice were conditionally depleted of β2-microglobulin expression in CD11c+ APCs. In these mice, no OT-I cell priming occurred in response to Abelson-mOVA tumor, which requires cross-presentation. However, priming was observed following OVA mRNA-LNP vaccination.
The researchers then generated BM chimeras by transferring WT or MHC-I triple KO donors into irradiated CD45.1+ recipient mice lacking NK cells. The chimera in which cDCs completely lacked self-MHC-I still generated antigen-specific CD8+ T cells after vaccination, suggesting MHC-I expression by cDCs was not required. To quantify the contribution of cross-dressing, BM chimeras were generated by transferring WT or cDC1-deficient BM into SJL or MHC-I TKO recipient mice. In WT-to-TKO chimeras, cDCs cannot acquire peptide–MHC-I from non-hematopoietic cells, but remain capable of direct presentation and cross-presentation. These chimeras had reduced OT-I proliferation. Further, cDC1-deficient-to-TKO chimeras showed an even higher reduction, suggesting that cross-dressing significantly contributed to cDC2-induced CD8+ T cell priming after mRNA-LNP vaccination.
Recent work has shown that cross-dressing by cDC2s in tumors depends on type I IFN signaling, a pathway activated by mRNA vaccines. To confirm the requirement for type I IFN, cDC1-deficient mice were immunized with mRNA-LNP in the presence of an IFNAR1-blocking antibody, which reduced the number of induced specific CD8+ T cells. Further, this blockade also reduced transfer of H2-Kb to cDCs in itgax-cre+B2mfl/fl mice.
To test MHC-I transfer from non-hematopoietic cells to cDCs, allogeneic BM chimeras were generated between B6 (H-2b) and BALB/c (H-2d) mice, to allow detection of distinct H-2K alleles on donor-derived cDCs. cDC1 and cDC2 in the spleens of vaccinated BALB/c-to-B6 chimeras expressed high levels of H2-Kd and acquired low levels of H2-Kb. An H2-Kb-restricted CD8+ T cell response was generated after vaccination, supporting a cross-dressing model.
The researchers then assessed whether vaccination-induced CD8+ T cells were functional in a 1956-fibrosarcoma model expressing membrane-bound OVA (1956-mOVA), which requires cDC1-mediated priming of CD4+ and CD8+ T cells. WT or cDC1-deficient mice were vaccinated with OVA mRNA-LNP and inoculated with 1956-mOVA in the contralateral flank. Vaccination prevented tumor growth, suggesting that cDC2-primed CD8+ T cells have antitumor activity.
To investigate whether cDC1 and cDC2 generated distinct memory responses, memory CD8+ T cells were profiled in OVA mRNA-LNP-vaccinated WT, cDC1-deficient, and cDC2-deficient mice 5-6 weeks after the second dose. Generated OVA-specific CD8+ T cells contained a higher percentage of CD127+ cells (CD127 is a memory cell marker) in cDC1-deficient than in cDC2-deficient mice. Cells from each model induced in vitro killing, suggesting both cDC1 and cDC2 generate functional memory CD8+ T cells, although cDC2 generated a larger memory pool.
ScRNAseq and TCRseq analysis of OVA-specific CD8+ T cells isolated from these mice showed that cDC2-deficient mice had a higher proportion of cycling and stem-like populations, while WT and cDC1-deficient mice had more clonally expanded terminal effector cells.
Overall, this study shows that both cDC1s and cDC2s can act as APCs for CD8+ T cell priming in response to mRNA-LNP vaccination, with cross-dressing driving this effect. This cross-dressing could broaden the repertoire of antigens presented beyond those targeted by the vaccine, potentially improving antitumor efficacy.
Write-up by Maartje Wouters, image by Lauren Hitchings




