Triple-negative breast cancer (TNBC) is characterized by DNA repair deficiency, genomic instability, and an immunogenic tumor microenvironment, suggesting it might serve as a candidate for individualized vaccination targeting somatic tumor mutation-associated neoantigens. Sahin, Schmidt, et al. recently published the results of a first-in-human clinical study with this strategy in Nature.
To develop the individualized neoantigen RNA vaccine, next-generation sequencing of tumor tissue was used to call mutations and predict T cell immunity-inducing neoantigens. For each patient, up to 20 mutations were concatemerized and encoded on two strings of immunostimulatory, non-nucleoside-modified uridine mRNA. A secretion signal and an MHC-I trafficking domain tag were fused to the coding sequence, and the mRNA vaccine was formulated in liposomal nanoparticles.
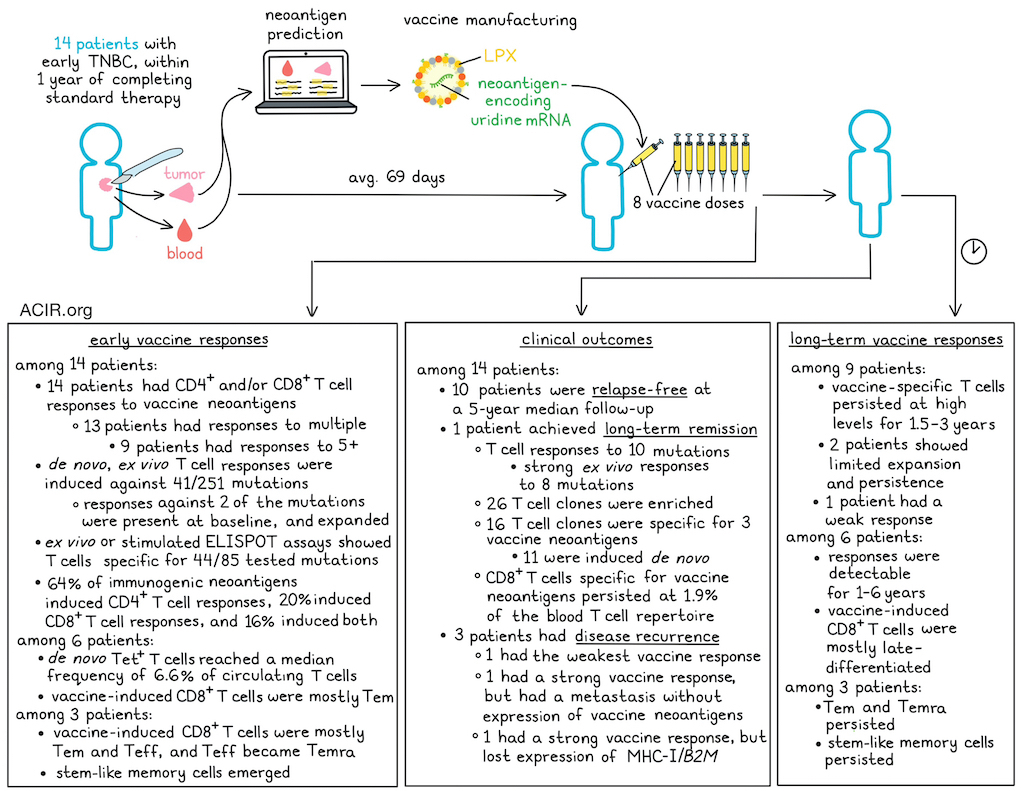
The clinical study included 14 patients who were within one year after completing standard therapy for early-stage TNBC. Patients received 8 doses of the vaccine. The first 3 patients started at a 14.4ug dose, escalating to the 50ug target dose, and received an off-the-shelf RNA-LPX-based vaccine encoding shared tumor-associated self-antigens to bridge the period of vaccine manufacturing. The next 11 patients received a 50ug vaccine dose from the start, without a bridge.
Vaccine manufacturing was feasible in a standard clinical setting, with an average turnaround time of 69 days from tumor sample to vaccine release. The most frequently reported treatment-emergent adverse events were reactogenicity symptoms, which occurred within 3 days, were mostly grade 1-2 and manageable, and resolved within a day.
To test for neoantigen-specific T cell responses, blood obtained at baseline and 7–14 days after the final vaccine dose was subjected to two IFNγ ELISPOT assays: an ex vivo assay using PBMCs incubated with overlapping peptides (OLPs) spanning the sequences of the neoantigens included in each vaccine, and a second assay after in vitro stimulation (IVS) of CD4+ and CD8+ T cells with autologous OLP-loaded APCs. All 14 patients had vaccine-induced or -amplified T cell responses against 1–10 of their vaccine antigens; 13 had responses directed against multiple antigens, with 9 having responses against ≥5 neoantigens. T cells specific for 41/251 total mutations across the 14 patients were detected de novo after vaccination; T cells targeting 2 neoantigens were present at baseline, and expanded following vaccination. The post-IVS ELISpot assay showed specific T cells for 44 of 85 tested mutations, of which over half were induced de novo. Approximately 64% of immunogenic neoepitopes were recognized by CD4+ T cells, 20% by CD8+ T cells, and 16% by both CD4+ and CD8+ T cells.
For 9 patients, the durability of vaccine-induced immune responses could be assessed using ex vivo ELISPOT with samples obtained during long-term follow-up. This showed that vaccine-specific T cells underwent a rapid expansion, followed by a brief contraction, and were then maintained at high levels over 1-3.5 years. However, in two patients, expansion and persistence were limited, and in one patient, responses were very weak.
Using 10 peptide:MHC-specific tetramers across 6 patients, de novo-induced, mutation-specific CD8+ T cells expanded quickly during the vaccination stage, becoming detectable after 3 doses and reaching a median frequency of 6.6% of all circulating CD8+ T cells after the final dose. Several of these specific CD8+ T cells remained detectable in the periphery for 1–6 years. Vaccine-induced CD8+ T cells were predominantly of an effector memory (Tem) phenotype in the early months, after which they acquired a late-differentiated phenotype that persisted for years.
One patient achieved long-term remission (>6 years), and blood was collected throughout this period. In this patient, T cell responses were detected against 10 of the vaccine-included mutations, with strong ex vivo ELISPOT responses to 8 neoantigens; the other 2 responses were weaker and mediated by CD4+ T cells. Further assessing this patient’s T cell repertoire, a combination of bulk and single-cell TCR sequencing was performed to detect neoantigen-specific clonotypes. The researchers assessed 26 CD8+ T cell clones that were most enriched post-vaccination; 16 recognized 3 ELIPOT-positive vaccine targets, of which 11 were induced de novo after vaccination. Neoantigen-specific T cells persisted at approximately 1.9% of the CD8+ TCR repertoire for >6 years in this patient.
scRNAseq and TCRseq data from CD8+ T cells from 3 patients showed that after the final vaccination dose, most neoantigen-specific CD8+ T cells had a terminal effector memory re-expressing CD45RA (Temra) phenotype, which persisted throughout follow-up. Early responses at the end of treatment were mostly defined by effector memory (Tem) and effector (Teff) T cells. Over time, Teff were replaced by Temra cells, which, together with Tem, became the dominant subtype during long-term follow-up. Further, stem-like memory (Tsm) populations emerged and persisted.
Of the 14 patients, 10 remained relapse-free at a median follow-up of 5 years, and one patient remained relapse-free until death from unknown causes. Three patients experienced a disease recurrence. Of these, one patient had the weakest vaccine-induced response, and the recurrence occurred 20 months after the last dose. The new tumor lesion contained T cells, and tumor cells expressed MHC-I. The patient was treated with anti-PD-1 and chemotherapy and had a complete response, lasting 15 months, after which the patient died due to systemic relapse.
The other two patients with recurrences had strong vaccine-induced immune responses against multiple vaccine targets. The first patient had bilateral BRCA1-mutated TNBC, and the vaccine was based on one of the two tumors. The metastasis retained MHC expression, but no somatic mutations from the tumor used for the vaccine creation were present, and no neoantigen-specific TCRs were detected in the metastasis. The second patient also had a good vaccine response, with 10 CD8+ clones specific for 4/6 vaccine neoantigens. However, while the relapse expressed most of the neoantigens in the vaccine, there was a loss of MHC-I and B2M gene expression in tumor cells, suggestive of downregulation of antigen presentation and immune escape.
The data from this first-in-human study suggest that this mRNA vaccine approach is feasible and efficient in inducing strong, long-lasting antigen-specific T cell responses. Research into mechanisms behind relapses after vaccination may inform future studies into combination therapeutic strategies or new targets for subsequent treatments to improve clinical outcomes.
Write-up by Maartje Wouters, image by Lauren Hitchings




