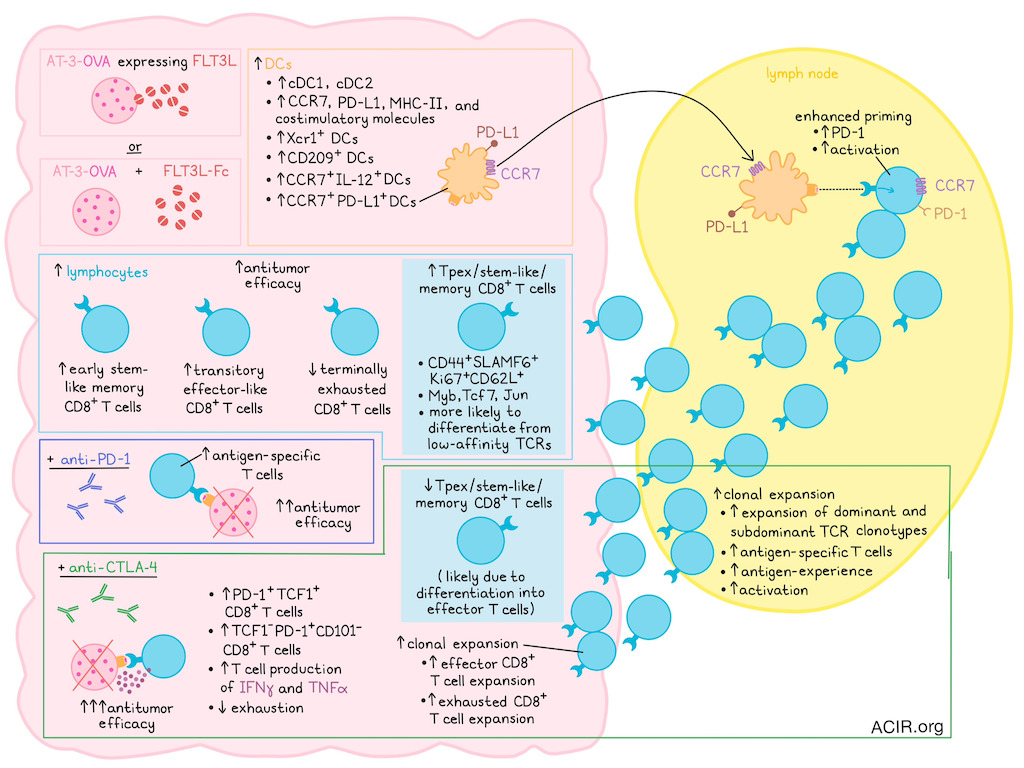
An important predictor of response to immune checkpoint blockade (ICB) is the presence of precursor exhausted T cells (Tpex) in the tumor. Conventional type 1 DCs (cDC1) are known to maintain these Tpex, and modulation of these factors might promote better therapeutic responses. In a recent publication in Nature Immunology, Lai, Chan, Armitage, et al. investigated whether Flt3L treatment could promote cDC1s and Tpex.
The researchers started by assessing the impact of Flt3L overexpression by tumor cells on immune cells using RNAseq. Flt3L overexpression increased the proportions of lymphocyte and DC clusters, with three enriched DC clusters characterized by high expression of Xcr1, Cd209, or Ccr7/Il12. In the lymphocyte compartment, a stem-like CD8 population expanded. Pseudotime trajectory analysis showed that ~50% of CD8+ T cells were in an early stem-like/memory state in Flt3L+ tumors, compared with ~20% in controls.
Mice with Flt3L-overexpressing AT-3-OVA tumors had a higher proportion of cDC1 and DCs expressing increased levels of CCR7, PD-L1, costimulatory molecules, and MHC-II. CD8+ T cells in these tumors had a less terminally exhausted phenotype, with an increase in CX3CR1+TCF1- cells, resembling transitory effector cells. There was also an increase in CD44+CD62L+SLAMF6+Ki67+CD8+ T cells, suggestive of a Tpex population with stem-like potential.
Given that recombinant FLT3L has been developed as a tumor therapeutic, AT-3 OVA tumor-bearing mice were then treated by intraperitoneal injection of recombinant Flt3L-Fc. Similarly to Flt3L overexpression, this treatment resulted in increased tumoral cDC1, cDC2, CCR7+PD-L1+ DCs, and CD62L+SLAMF6+Ki67+CD8+ T cells. Using an XCR1-DTR mouse model in which diphtheria toxin treatment depletes tumor cDC1 and partially reduces CCR7+PD-L1+ DCs, the researchers showed that these DCs are required for the increase in tumoral CD62L+SLAMF6+Ki67+CD8+ T cells.
Further transcriptional analysis of these stem-like/memory CD8+ T cells induced by Flt3L revealed increased expression of the transcription factors Tcf7, Myb, and Jun. In particular, Myb was shown to be important for this phenotype, as Myb knockout in OT-1 cells reduced the formation of CD62L+SLAMF6+CD8+ T cells after Flt3L treatment.
Given that CD8 memory formation is linked to T cell receptor (TCR) signal strength, the researchers then assessed the relationship between CD62L+SLAMF6+CD8+ T cell differentiation and TCR signal strength. When OT-I (high-avidity TCR) and OT-III (low-avidity TCR) cells were transferred into Flt3L-conditioned mice, a higher proportion of CD62L+SLAMF6+ cells was detected among the OT-III cells, suggesting that T cells with low-avidity TCRs are more likely to differentiate into the Tpex phenotype.
To assess the role of the draining lymph node (dLN) in the increase in the CD62L+SLAMF6+ T cell population, a clustering analysis of LN and tumoral T cells was performed. Tumor-infiltrating CD62L+SLAMF6+ cells clustered closely to dLN central memory T cells (Tcm), and were distinct from tumoral Tex, dLN effector memory T cells (Tem), and naive T cells. When mice were treated with FTY-720 to block T cell lymph node egress, the number of CD62L+SLAMF6+CD8+ T cells decreased, suggesting that the dLN is required for the induction of this population.
Knockout of CCR7 on OT-I cells before transfer into mice removed the effects of Flt3L, indicating that Flt3L treatment may induce DC differentiation that requires migration of tumor antigen-specific T cells in the dLN for priming. To test this hypothesis, the researchers compared OT-I phenotypes in dLNs and found higher PD-1 expression in mice bearing Flt3L+ tumors, suggesting priming had occurred. When OT-I cells were engineered to express GFP downstream of the NR4A2 promoter (a gene activated immediately downstream of TCR activation), higher GFP levels were detected in the dLNs of mice with Flt3L-expressing tumors, confirming enhanced priming.
CCR7 deletion in myeloid cells in a bone marrow chimera system reduced DC numbers in the dLN and CD62L+SLAMF6+CD8+ T cells in the tumor, suggesting the importance of not only T cell migration, but also DC migration to the LN for the formation of this population.
In Flt3L-expressing AT-3 OVA tumors, the antitumor immune response further improved following anti-PD-L1 or anti-CTLA-4 treatment. ICB increased antigen-specific T cells in the tumor, with effects most pronounced in the CTLA-4-treated groups. While Flt3L led to an increase in CD62L+TCF1+CD8+ T cells, adding anti-CTLA-4 resulted in a decrease of such cells, which may be due to increased differentiation of these cells into effector cells. This was confirmed by increased PD-1 levels on CD62L+TCF1+ cells, the presence of more TCF1-PD-1+CD101- T cells, and increased IFNγ and TNF production by T cells. Bulk TCRseq of LNs following treatment revealed expansion of dominant and subdominant TCR clonotypes, and this clonotypic expansion was inhibited by FTY-720 treatment.
scRNAseq and TCRseq analysis showed that the combination of Flt3L and anti-CTLA-4 led to the emergence of clonally expanded CD8+ T cell populations in the dLN, with increased expression of genes associated with antigen-experienced T cells and T cell activation. In the tumor, Flt3L increased the proportion of stem-like cells and led to a strong enrichment of genes associated with the Myb-dependent Tpex population. There was more clonal expansion among clusters of effector-like T cells than among clusters with stem-like cells. Adding anti-CTLA-4 treatment resulted in expanded CD8+ effector/Tex cell populations.
CTLA-4 blockade, in the context of Flt3L, expanded a cluster of genes related to stem-like/memory and effector-like populations, while reducing genes related to exhaustion. Pseudotime analysis suggested this population was an intermediate population between the Tpex and Tex clusters, and the cells shared TCRs with effector and stem-like clusters.
Comparing the derived gene signatures in the above analyses to datasets of patients undergoing ICB treatment. Patients had better therapeutic responses when an Flt3L gene signature was expressed, and genes related to a cluster that expanded upon Flt3L treatment were associated with improved survival.
Together, these data suggest that Flt3L supports DC priming of antitumor immune responses, leading to a less differentiated, stem-like population of CD8+ Tpex cells that is more responsive to ICB. Therefore, this combination treatment might help induce an antitumor response in tumors that are currently not responding to ICB.
Write-up by Maartje Wouters, image by Lauren Hitchings.




