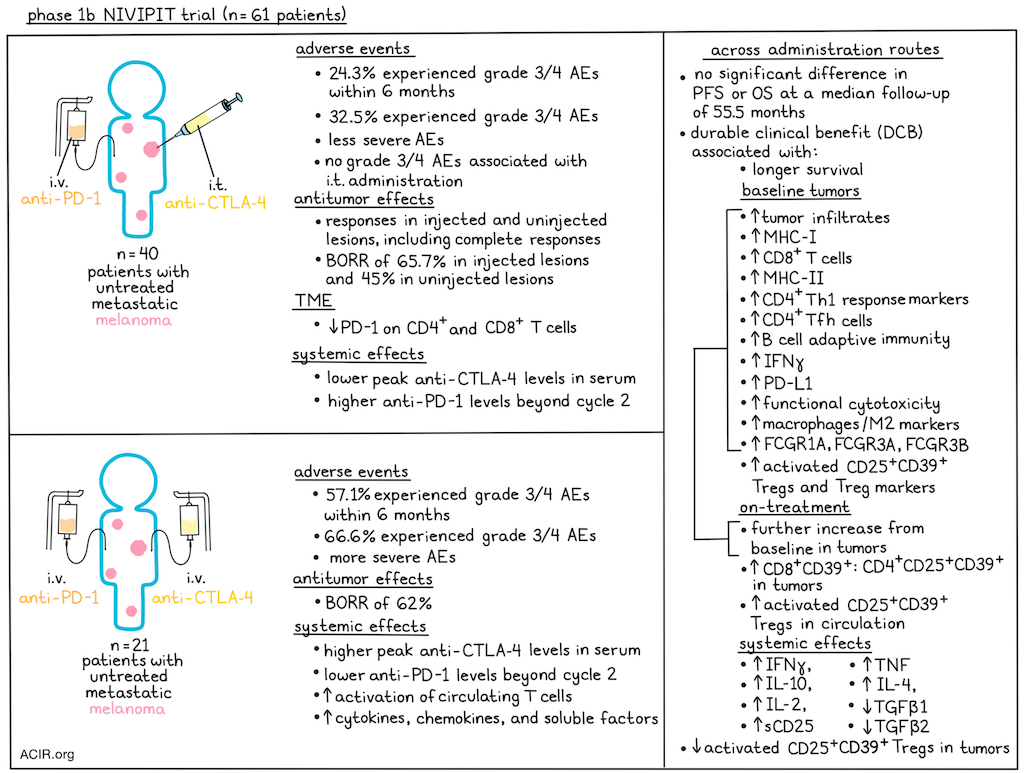
The combination of nivolumab (anti-PD-1) and ipilimumab (anti-CTLA-4), both administered intravenously, has been approved for the treatment of cancer in a variety of settings since 2015, but its use is frequently hindered by severe adverse events, more often associated with the use of anti-CTLA-4. In an effort to reduce this toxicity, Tselikas and Susini et al. conducted a randomized, multicenter, phase 1b clinical trial (NIVIPIT) in which 61 patients with untreated metastatic melanoma were treated with intravenous anti-PD-1 in combination with either intravenous (i.v.) or intratumoral (i.t.) ipilimumab (anti-CTLA-4). Their results were recently published in Nature.
In the NIVIPIT trial, the 61 patients enrolled across four treatment sites were randomized at a 2:1 ratio to receive either i.v. anti-PD-1 and i.t. anti-CTLA-4 (n=40) or the standard i.v. anti-PD-1 and i.v. anti-CTLA-4. Patients were treated with both anti-PD-1 and anti-CTLA-4 every 3 weeks for 4 doses, followed by treatment with just anti-PD-1 every 2 weeks for up to 12 months. In the i.t. anti-CTLA-4 group, 162 injections were performed across 46 tumor lesions, most of which were lymph node, cutaneous, or subcutaneous metastases, though some deeper lesions (liver and lung) were also injected.
The main objective of the study was to evaluate 6-month treatment tolerance. Among the 37 evaluable patients in the i.t. treatment arm, 9 patients (24.3%) experienced a treatment-related grade 3 or 4 adverse event within 6 months, which was below the pre-specified threshold of 30%, and less than the 57.1% patients in the i.v. arm who experienced treatment-related grade 3 or 4 adverse event within 6 months. Over the duration of the follow-up, the cumulative incidence of grade 3 or 4 treatment-related adverse events was 32.5% in the i.t. arm and 66.6% in the i.v. arm. AEs were also generally less severe in the i.t. arm, where only one grade 4 event was reported. No grade 3 or 4 AEs related to the intratumoral injection procedure were reported.
Looking at the concentrations of anti-PD-1 and anti-CTLA-4 in patient serum, the researchers noted that peak levels of anti-CTLA-4 were significantly lower in the i.t. arm, while anti-PD-1 was unexpectedly lower beyond cycle 2 in patients in the i.v. arm.
Evaluating antitumor activity in the i.t. arm, the researchers observed notable responses in injected lesions, with several instances of complete responses. These responses were often accompanied by responses in distant uninjected lesions, consistent with a systemic immune response. In the i.t. arm, the best overall response rate (BORR) was 65.7% in injected lesions and 45% in uninjected lesions. In the i.v. arm, the BORR was 62%. Complete responses were observed in a higher proportion of injected lesions in the i.t. arm than overall in the lesions analyzed in the i.v. arm. After a median follow-up of 55.5 months, there were no statistically significant differences in progression-free or overall survival between treatment arms, though it is important to note that this trial was not powered for formal comparisons between arms. Durable clinical benefit (DCB; defined as maintenance of RECIST response or stable disease beyond 6 months) was associated with prolonged survival, regardless of the route of anti-CTLA-4 administration.
Next, Tselikas and Susini et al. evaluated the pharmacodynamic effects of treatment and found that i.t. anti-CTLA-4 administration reduced PD-1 expression on CD4+ and CD8+ tumor-infiltrating lymphocytes in injected lesions while also inducing systemic pharmacodynamic effects typically observed with i.v. anti-CTLA-4. With i.v. doses of anti-CTLA-4, which resulted in higher circulating anti-CTLA-4 levels than i.t. dosing, the researchers also noted increased activation markers on circulating T cells and higher serum levels of a variety of cytokines, chemokines, and soluble factors, including IL-2, soluble CD25 (IL-2Rα), IL-12/IL-23p40, IL-27, IL-17A, VEGF, and soluble PD-L1, suggestive of increased systemic immune activation. Increased systemic IFNγ, IL-10, IL-2, sCD25, IL-4, and TNF, and decreased systemic TGFβ1 and TGFβ2 were associated with DCB across both the i.t. and i.v. arms.
Looking at tumor features that might be associated with responses, the researchers found no significant difference in tumor mutational burden or somatic copy-number alterations between patients with and without DCB. Tumor infiltrates, MHC-I, CD8+ T cells, MHC-II, markers of T helper 1 CD4+ T cell responses, Follicular helper T (TFH) cells, B cell-mediated adaptive immunity, IFNγ, PD-L1, and evidence of functional cytotoxicity were all higher at baseline in patients with subsequent DCB. A number of these pre-existing immune features increased quantitatively three weeks after treatment initiation in patients with subsequent DCB, but not in those without.
Interestingly, in addition to increased markers associated with pre-existing MHC-I- and MHC-II-dependent adaptive antitumor immunity, patients with DCB also showed increased levels of a previously reported activated intratumoral Treg population (CD25+CD39+) and Treg-associated markers, including Foxp3, CD25, CCR8, IL-10 and CTLA-4. This population of intratumoral activated Tregs significantly decreased upon treatment. In patients with DCB, circulating Tregs increased, and there was a trend towards increased CD8+ T cell:Treg ratios in tumors. In patients without DCB, membrane protein expression of CTLA-4 increased within the same cell population of intratumoral activated Tregs, and the CD8+ T cell:Treg ratios remained unchanged.
Investigating the potential role of FcγR-mediated Treg cell depletion by anti-CTLA-4, the researchers found that expression of FCGR1A, FCGR3A, and FCGR3B was higher at baseline in tumors from patients with DCB, and increased further upon treatment. Macrophages and markers of an M2-like state were also more abundant at baseline in patients with DCB, and increased upon treatment, but not in patients without DCB. These intratumoral features were similar between administration routes.
Overall, these results suggest that intratumoral anti-CTLA-4 combined with intravenous anti-PD-1 is safe in advanced melanoma, with a substantial reduction in the incidence of severe treatment-related adverse events compared to intravenous administration, no severe adverse events related to the administration route, and a high frequency of complete responses in injected lesions. While intratumoral anti-CTLA-4 induced less systemic activation than intravenous, it still induced systemic changes and an abscopal effect, yielding responses in both injected and uninjected lesions. Additionally, this research showed that high pre-existing adaptive immunity as well as high CD25+CD39+ Tregs and (perhaps mechanistically linked) high levels of macrophages expressing FcγR in tumors could predict DCB from combination anti-PD-1 and anti-CTLA-4, regardless of the route of administration.
Write-up and image by Lauren Hitchings
Meet the researcher
This week, co-first author Sandrine Susini answered our questions.

What was the most surprising finding of this study for you?
The most surprising finding was the predictive power of activated intratumoural regulatory T cells and M2 macrophages at baseline, as these were historically considered purely immunosuppressive. Specifically, we observed that a high abundance of activated intratumoural Tregs and M2 macrophages before treatment actually predicted a durable clinical benefit. A major challenge was overcoming the controversy regarding whether anti-CTLA-4 actually depletes Tregs in humans. We succeeded by using fresh tumor biopsies, which allowed us to identify that this depletion is highly selective for the activated subset of Tregs, rather than the total population.
What is the outlook?
The outlook for this research is highly promising, particularly regarding its potential as a game-changer for patient safety. We have a strong rationale to move this strategy into earlier tumor settings. We are looking toward neoadjuvant or oligometastatic contexts, where local control is critical, with minimal side effects. Finally, the translatability of our work is enhanced by the use of fresh tumor analysis. Techniques like flow cytometry and secretome analysis can provide rapid, biologically grounded biomarkers to forecast therapeutic benefits within hours, offering a path toward truly personalized immunotherapy.
Who or what has been a major source of inspiration or motivation for you throughout your career?
Throughout my career, one of my strongest sources of motivation has been the feeling of being useful. Knowing that our research can ultimately contribute to improving treatments for patients is incredibly meaningful to me, and gives real purpose to my work.




