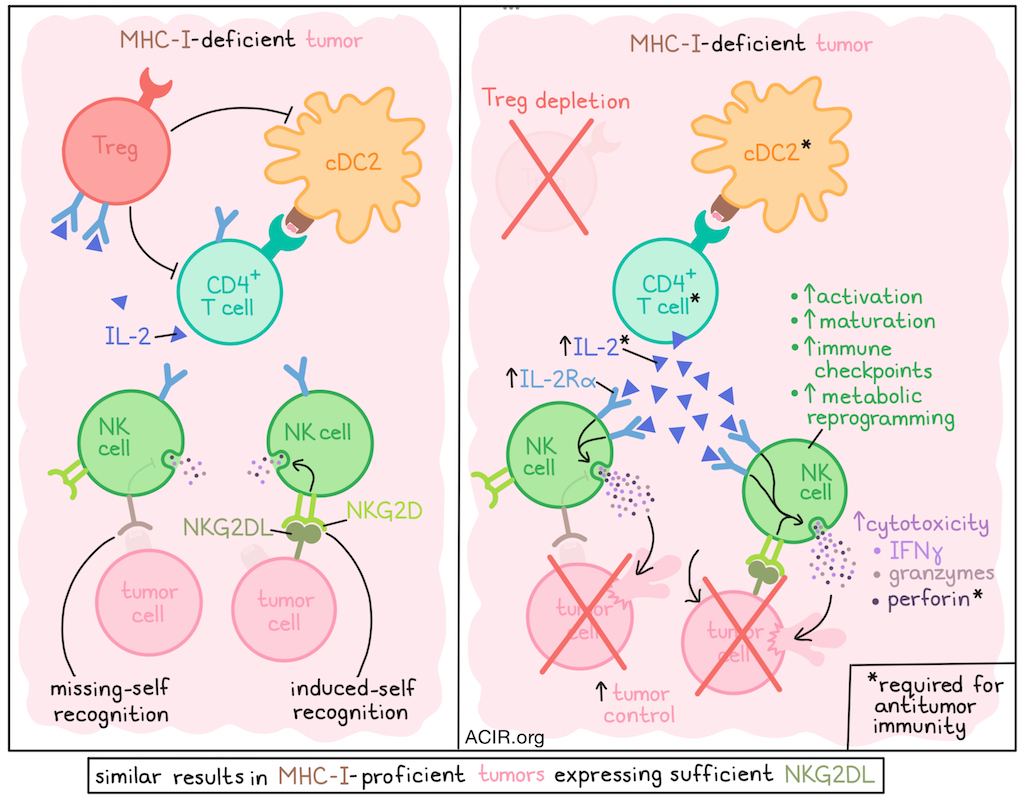
When tumors lose expression of MHC-I under the selective pressure of CD8+ T cells, NK cells may step in and take on a more prominent antitumor role, but often this is still insufficient to control tumors. Investigating potential mechanisms limiting NK cell antitumor activity, Zhang et al. found that Tregs have a strong inhibitory effect on NK cells. Intratumoral Treg depletion could unleash increased NK cell functionality against MHC-I-deficient, and even MHC-I-proficient tumors, dependent on cDC2s and conventional CD4+ T cell (Tconv) production of IL-2. Their results were recently published in Science Immunology.
To begin, Zhang et al. established MHC-I-deficient MC38, B16F10, and RMA lymphoma tumors in Foxp3 DT-GFP mice, which could be depleted of Tregs upon administration of diphtheria toxin (DT). In each of the three tumor models, Treg depletion induced tumor control, but also initiated autoimmune toxicity that required eventual euthenasia. This toxicity could be prevented by administering DT intratumorally, avoiding systemic T cell activation.
To identify which cell types mediated tumor rejection after Treg depletion, the researchers co-depleted other immune cells and found that while CD8+ T cells did not impact Treg depletion-mediated tumor control, CD4+ Tconv and NK cells were both essential. After Treg depletion, CD4+ Tconv were increased only in some models, while NK cells were not increased, suggesting functional changes were primarily responsible for the antitumor effects, rather than proliferation or recruitment of these cell types.
Using an RMA-H60a lymphoma model, which naturally expresses MHC-I and was engineered to express high levels of the activating NKG2D ligand H60a, the researchers were able to show that the antitumor effect of Treg depletion was dependent on the presence of the H60a, even in the presence of MHC-I, demonstrating NK cell activity could be enhanced through both “missing self” (missing MHC-I) and “induced self” (presence of NK cell ligands) recognition of target cells.
Investigating functional changes in NK cells after Treg depletion, the researchers used bulk RNAseq to profile NK cells from MHC-I-deficient MC38 tumor models and found that 6 days after Treg depletion, genes associated with activation, immune checkpoints, and cytotoxicity were upregulated, including genes encoding granzyme A, granzyme B, perforin, and orphan granzymes (C–N). Treg depletion also altered NK cell metabolic pathways, favoring pathways consistent with an oxidative and/or hypoxic stress response, and a general inflammatory state. These transcriptional changes were largely dependent on the presence of CD4+ Tconv.
To validate their transcriptional results at the protein level, Zhang et al. performed flow cytometry on tumor NK cells and found that by day 4 after Treg depletion, they were more proliferative (BrdU labeling) and expressed higher levels of the early activation marker CD69. By day 6, NK cells also upregulated activation markers SCA-1 and KLRG1, exhibited a more mature phenotype, (CD11b+CD27−), had increased intracellular granzyme B and perforin, and demonstrated increased ex vivo cytotoxicity. NK cells also showed increased IFNγ production with or without ex vivo stimulation, suggesting that some stimulation had occurred in vivo. These phenotypic changes were dependent on CD4+ Tconv, and tumor control was dependent on perforin, though degranulation (CD107a) was not increased, suggesting an increase in NK cell cytotoxic potential. Similar phenotypic changes in NK cells were also observed in the RMA-H60a model.
Given that Tregs have been shown to regulate the activation and priming of effector T cells both directly and indirectly through suppression of DCs, the researchers investigated whether DCs were essential to the antitumor effects of Treg ablation. Using models lacking cDC1s, cDC2s, or both, the team showed that cDC2s were essential to the antitumor effects.
Gene set enrichment analysis of tumor NK cells after Treg depletion showed enrichment of several cytokine signaling pathways, including IL-2–STAT5 signaling, TNF-α signaling via NF-κB, and interferon-gamma response pathways, all of which were largely unenriched when CD4 Tconv cells were depleted. Investigating the IL-2–STAT5 signaling pathway, the researchers found that NK cells in tumor, but not spleens or dLNs, expressed increased cell surface IL-2Rα (CD25) and increased STAT5 activation after Treg depletion, suggesting local IL-2-mediated activation. Given that Tregs can limit CD4+ Tconv activation both by suppressing CD4+ Tconv IL-2 production and by acting as an IL-2 sink due to high surface expression of IL-2Rα, the researchers hypothesized that this mechanism also affected NK cells. Indeed, the team confirmed that CD4+ Tconv increased IL-2 production upon Treg depletion, and CD69 and KLRG1 were upregulated. These effects were dependent on cDC2s.
To determine the effects of IL-2 availability on NK cell functionality, Zhang et al. treated tumor-bearing mince with an IL-2-neutralizing antibody at the time of Treg ablation, and found that this largely prevented changes in NK cell functionality and NK cell-mediated tumor control. Neutralizing TNFα or IFNγ did not have a similar impact, highlighting the importance of IL-2 availability. Further, administration of exogenous half-life-extended IL-2 to tumor-bearing mice that were not depleted of Tregs also promoted tumor control, which was largely abrogated when NK cells were depleted.
Investigating clinically relevant Treg depletion options, the researchers confirmed high CCR8 expression on Tregs in MHC-I-deficient tumors and administered anti-CCR8 systemically to deplete Tregs. This resulted in control of established tumors that were dependent on NK cells and CD4+ Tconv, but not CD8+ T cells, and was associated with increased expression of KLRG1 and SCA-1 on NK cells. Similar tumor control was observed with DT-mediated Treg ablation or anti-CCR8-mediated Treg depletion in two orthotopic tumor models.
Overall, these results suggest that Tregs depletion could sensitize both MHC-I-deficient and MHC-I-proficient tumors to NK cell-mediated cytotoxicity, which required cDC2-mediated activation of CD4+ Tconv and subsequent production of IL-2. Increasing IL-2 availability could also enhance NK cell antitumor responses, supporting several potential clinical avenues for immunotherapy that involve either eliminating Tregs or supporting NK cells.
Write-up and image by Lauren Hitchings
Meet the researcher
This week, first author Chenyu Zhang answered our questions.

What was the most surprising finding of this study for you?
We found that the tumor-specific NK cell response unleashed by Treg depletion was novel and unique, as it required help from CD4+ T cells, just as B cell and memory CD8+ T cell responses do.
What is the outlook?
We were surprised at the critical role of CD4+ T cells in the control of MHC I-deficient tumors, especially in driving a strong NK cell antitumor responses, but also independently of NK cells and CD8+ T cells. We will pursue the analysis of the independent role of CD4+ T cells in further studies.
If you could go back in time and give your early-career self one piece of advice for navigating a scientific career, what would it be?
Comparison is the thief of success – just aim to do the best and most rigorous science you can!




